Search the Community
Showing results for tags 'analysis'.
-
Having a think.... I have fifty non-homogeonous biological samples I want to send to a NATA accredited lab for analysis. They contain volatiles, so heating the samples will drive off some of the compounds I want to check the levels off. Heating the samples is part of the standard process of digesting them so they can be analysed. Freeze drying is an option, but for that number of samples the prep would be expensive and out of my range The compounds I'm looking at are relatively stable at ambient temperatures, or at least any variations for the more volatile compounds which come off at above 60C aren't compounds of interest. I was thinking of grinding samples to dryness under liquid nitrogen in a not-fully-sealed stainless lab blender- because I have one here ( or should I seal it? ) but was told that as soon as the liquid nitrogen evaporates , water will rush in and the sample won't be dry any more. Is this true? How fast does the water rush back in if I quickly place the dried samples into a -20 freezer I've previously dried and ground biological samples under liquid nitrogen and found that most suitable, but that was for DNA analysis, not chem analysis. Next option is to grind and dry at 50C under air. Neither will give me consistent moisture levels for all samples, and it will prolly be up to the NATA lab to standardise these before analysis Does anyone reckon grinding under liquid nitrogen to be the superior method in this case? Ta for help, this place is fulla smarties :D
-
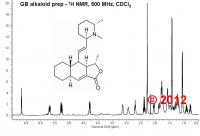
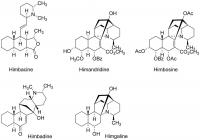
The genus Galbulimima (Himantandraceae) consists of large rainforest trees distributed in Australia (North Queensland), Papua New Guinea, and the Moluccas. Depending on the botanical authority 1-4 different species were acknowledged, but Australian G. baccata and New Guinean G. belgraveana are most often accepted. Previous reports have indicated that in Papua New Guinea the bark of G. belgraveana was ingested by specialized diviners to receive dreams of prediction (Hamilton 1960, Trans. PNG Sci. Soc. 1, 16-18). Self experiments have previously been described here: http://www.shaman-au...showtopic=19895 Although more than 28 alkaloids have been isolated from the bark so far (Brown et al. (1956) Aust. J. Chem. 9, 283-287; Binns et al. (1965) Aust. J. Chem. 18, 569-573) the active principle has remained unidentified. I became interested in Galbulimima belgraveana in 1994 while traveling in Papua Niugini. During extensive fieldwork in remote areas of the Eastern Highlands in 1999 and 2004 I managed to collect several samples under the supervision of practicing shamans who also introduced me into eating the bark (it can be VERY active). Fore shaman holding Agara bark (G. belgraveana) taken from a tree considered to be inactive because of its location in a recently cleared garden site. What might be particularly interesting with respect to the (ethno)botany of Galbulimima is that shamans differentiate between two kinds of trees, described as “man” & “meri” (male and female) although the trees are clearly monoecious. Furthermore, shamans employ a rigorous selection procedure to identify trees suitable for the preparation of the bark drug which takes morphological, ecological as well as supernatural considerations into account. Only trees from the primary rainforest at the top of mountain slopes are employed and those in areas of “ples masalai” (spirit places) are considered to be particularly powerful. It is generally assumed that Galbulimima trees exposed to frequent human contact loose their power because they are abandoned by the tree spirit who is believed to reside in them. Most interestingly, shamans selection procedures seem to be paralleled by an extreme variability in alkaloid composition observed during chemical analysis: “Whether the genus consists of one, two, three, or four species the variability of alkaloid content with respect both to total amount and to individual alkaloids is striking and possibly unique. Of the eleven bark samples from North Queensland and the seven from New Guinea that have been worked up for alkaloids no two have been alike. Even samples collected in the same area and at approximately the same season have been quite dissimilar. The variability thus revealed by actual isolation of the alkaloids has been confirmed by a paper-chromatographic survey of the alkaloids of an extensive series of samples. The yield of total alkaloids varied from a trace to about 0.5%. The major and commonest alkaloids in bark from both sources were himbacine and himandridine. Himbadine and himgaline, which are structurally related, were also major alkaloids in bark from North Queensland but were not found in material from New Guinea, except in one instance when a trace of himgaline was isolated. Himbosine was usually present in small amounts in all samples but all the other alkaloids occurred irregularly and should be regarded as minor. Some have been isolated only once.” Ritchie & Taylor (1967) in: The Alkaloids Vol.9, RHF Manske (Ed.), 529-543. Major alkaloids from New Guinean G. belgraveana and Australian G. baccata. Recently, I regained the opportunity to perform chemical analyses using state of the art analytical techniques (NMR and HPLC-MS) and have started to analyze my samples that had been stored away for many years at -20 °C. For isolation of crude Galbulimima bark alkaloids I employed the following extraction procedure: 1 g of dried bark was powdered and extracted with 3 x 10 ml methanol for 24 h at room temperature. The filtered extract was concentrated to dryness in vacuum and the residue taken up in 1 ml 5% aqueous hydrochloric acid and extracted with 3 x 1 ml diethyl ether. The aqueous phase was basified with 1 ml 20% aqueous ammonia solution and extracted with 3 x 1 ml chloroform. The organic phase was dried over sodium sulfate, filtered and concentrated to dryness in vacuum. The residue was taken up in 600 µl chloroform-d1 and analyzed by NMR spectroscopy. The 500 MHz proton NMR spectrum suggested that the crude alkaloid extract consists of almost pure Himgravine (approx. >90%). Isolated amounts exceed 10 mg/g of dried bark, which means that alkaloid content in this sample is at least twice as high as those reported in the literature. 1H NMR spectrum (500 MHz, CDCl3) of crude G. belgraveana alkaloids shows Himgravine as the major component. However, as no NMR data of authentic Himgravine have ever been published (apart from a very low resolution 1H NMR in Abraham & Bernstein (1961) Aust. J. Chem. 14, 64-68), I have also acquired a H,H-COSY spectrum to confirm the tentative identification of Himgravine. Again, the data are in excellent agreement with the proposed identification of Himgravine. However, I intend to acquire additional NMR spectra (13C NMR, HMQC, HMBC, and NOESY) to unambiguously establish the structure assignment and will sure run HPLC-MS to get an idea about the minor components. In addition, I will continue to analyze my various other G. belgraveana samples (man & meri, active & inactive) and might also try to isolate some of the alkaloids in pure form. H,H-COSY spectrum (500 MHz, CDCl3) of crude G. belgraveana alkaloids shows Himgravine as the major component. Please keep in mind that the analyzed sample is derived from a tree that was not used for divination purposes, so I do not claim that Himgravine is an active component. However, I do hope that analysis of my other samples (some of which are surely active) will enable identification of the active principle(s). What I’m now interested in is to get access to additional samples of authentic Galbulimima bark from Papua Niugini and Australia. The idea is to analyze active as well as inactive specimen in order to find candidate compounds for the observed activity and thus to bypass bioassays with all the different isolated components, some of which might in fact be harmful (especially if taken on their own). Consequently, samples which have already been bioassayed with positive as well as negative results would of course be most helpful. Amounts as low as 1 g of dry bark could be sufficient to identify the major component(s). Crude alkaloid mixtures could be send back if requested. Please be advised that I will NOT perform extractions of large bark samples for you – my intention is chemical analysis only. I’m also not interested in analyzing anything else apart from Galbulimima (no need to ask, sorry) and I will perform microscopic analysis of bark samples prior to any extraction – samples other than Galbulimima would be discarded. For those of you who feel inspired to visit Papua Niugini on their own in order to acquire G. belgraveana, I would like to give you the following advice: Learning Tok Pisin (Pidgin, Neo-Melanesian) is absolutely essential because people capable of speaking English do not know anything about traditional customs and plant use (translators are not suitable). Keep in mind that you are most likely to fall prey to con-men who will try to deprive you of your personal belongings. Don’t expect to obtain any (authentic) samples during your first visit. Plan at least several weeks living with the indigenous people before a reliable level of trust has been established. Sampling of any tree requires the consent of village leaders and landowners. Keep in mind that Galbulimima is used to divine the cause of sickness, which is most often considered to be sorcery. Your interest in knowledge of sorcery and counter sorcery makes you vulnerable to serious sorcery accusations and might severely endanger your safety. Comply with quarantine regulations. Ingestion of any plant material might pose serious health risks including permanent psychological or physical damage, or even death. Please stay safe.
- 11 replies
-
- 7
-

-
- agara
- galbulimima
-
(and 3 more)
Tagged with: